/MomentInertia-56fd5a985f9b586195c6d7a0.jpg) 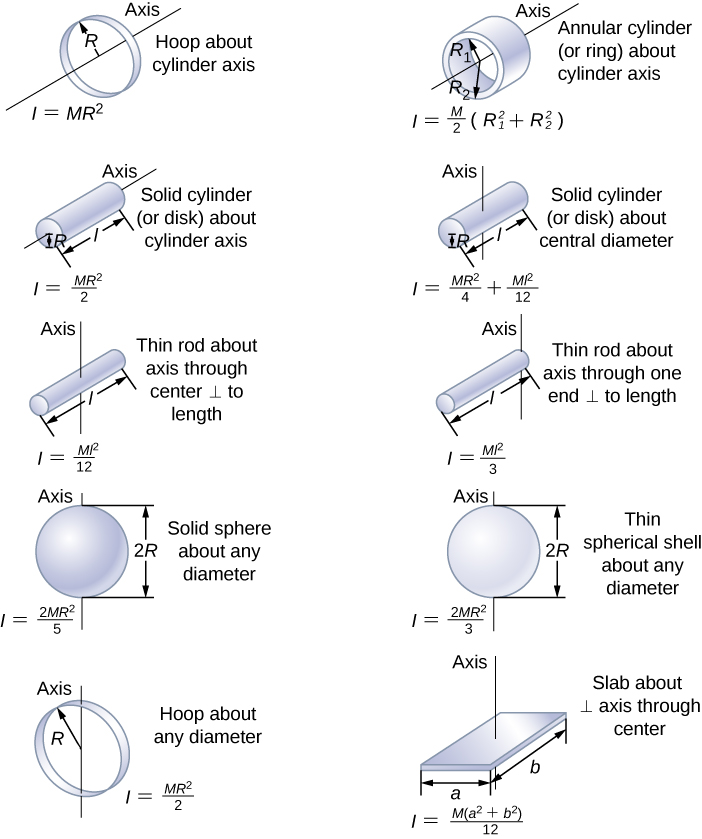
Here we have four obvious principal axes: only possible if What about a symmetrical three dimensional molecule? The moment of inertial about either of theseĪxes will be one-half that about the perpendicular-to-the-plane axis. Obviously, one principal axis is through the centroid,Įstablished that any axis of symmetry is a principal axis, so there areĮvidently three principal axes in the plane, one along each bond! The only interpretation is that there is aĭegeneracy: there are two equal-value principal axes in the plane, and any two Lowest angular momentum state E = L 2 / 2 I = ℏ 2 / 2 I , is lower for molecules with bigger moments of This was not the case for heavier diatomic gases, since the energy of the Was so low that a lot of energy was needed to excite the first quantized The resolution was that the moment of inertia 
Though they constantly collided with each other. Temperatures, apparently these diatomic molecules didn’t spin around, even Which gave an excellent account of the specific heats of almost all gases, 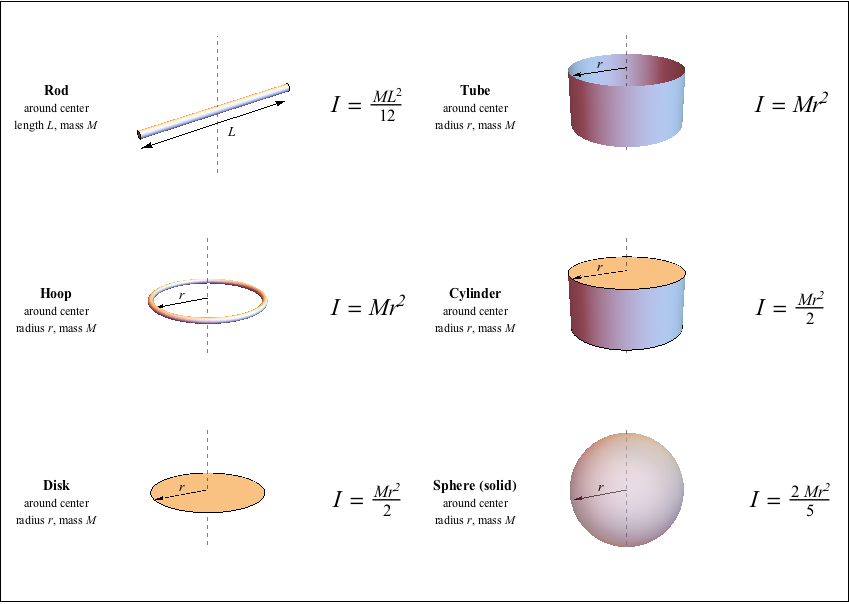
In the nineteenth century, the mystery was that equipartition of energy, Two point masses m a fixed distance a apart gives I = 1 2 m a 2. The moment of inertia of the hydrogen molecule wasįind: the nuclei (protons) have 99.95% of the mass, so a classical picture of Michael Fowler Examples of Moments of Inertia Molecules
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
